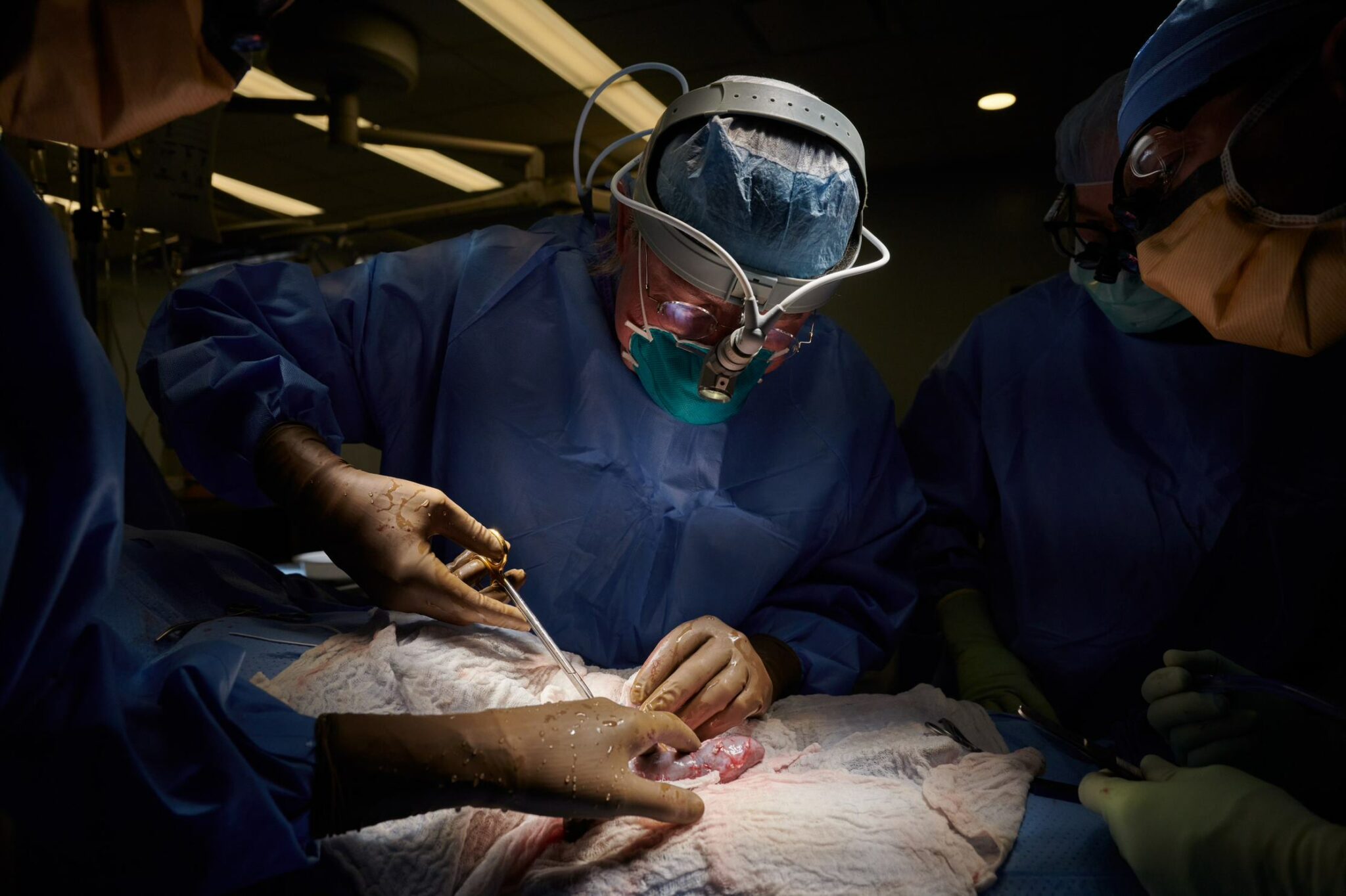
No, scientists are not any closer to pig-to-human transplants than they were last week
Steve Holtzman was awoken by a 1 a.m. call from a doctor at Duke University asking if he could put some pigs on a plane and fly them from Ohio to North Carolina that day. A motorcyclist had gotten into a horrific crash, the doctor explained. He believed the pigs’ livers, sutured onto the patient’s skin like an external filter, might be able to tide the young man over until a donor liver became available.
Unlock this article instantly by becoming a free subscriber.
You’ll get access to free articles each month, plus you can customize what newsletters get delivered to your inbox each week, including breaking news.